- Home
- Products
- Elementary
- Boride Powder
- 3D Printing Powder
- Sulfide Powder
- Oxide Powder
- Carbide powder
- Nitride Powder
- Silicide Powder
- Hydride Powder
- Telluride Powder
- Selenide Powder
- Stearic Acid Series
- Phosphide Powder
- Nanoparticles
- Metal Alloy
- MAX Phase
- Lithium Battery Anode
- Surfactant
- Molecular sieves
- Concrete Admixtures
- News
- Answers
- Contact
- About
News
- 1
- 1
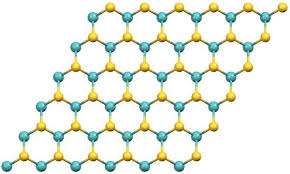
Zirconium diboride (ZrB2) :A remarkable ceramic material with a unique hexagonal crystal structure
If you are looking for high-quality products, please feel free to contact us and send an inquiry, email: brad@ihpa.net
Zirconium diboride (ZrB2) is a remarkable ceramic material with a unique hexagonal crystal structure. Its exceptional properties make it ideal for various applications, including aerospace engineering and high-performance electronics. With its superior strength, durability, and resistance to extreme temperatures, ZrB2 is a choice for those seeking the best-advanced materials. It is a refractory material with a high melting point and high hardness. Due to its exceptional properties, ZrB2 is a popular choice for various engineering applications. This article provides a comprehensive overview of zirconium diboride, including its properties, synthesis, applications, and future trends.
Properties of Zirconium Diboride
Zirconium diboride is a ceramic material with a hexagonal crystal structure. It has the following properties:
High melting point: ZrB2 has a high melting point of around 2700°C, making it suitable for high-temperature applications.
High hardness: ZrB2 has a hardness of around 1000–1500 HV, higher than many steels and tool alloys.
Lightweight: ZrB2 has a low density of around 3.4–3.5 g/cm³, making it an attractive material for light applications.
High electrical and thermal conductivity: ZrB2 has high electrical and thermal conductivity, making it suitable for electronic and heat-resistant applications.
High corrosion resistance: ZrB2 is highly resistant to acids, alkalis, and other chemicals.
ZrB2's low thermal expansion coefficient makes it suitable for thermal shock-resistant applications.

Synthesis of Zirconium Diboride
Zirconium diboride can be synthesized using various methods, including solid-state reaction, molten salt electrolysis, and gas-phase reaction. The most common way is solid-state reaction, which involves reacting zirconium oxide (ZrO2) and boron oxide (B2O3) at high temperatures. The reaction typically occurs in a vacuum or inert atmosphere to prevent oxidation of the reaction product. The reaction equation is: 3ZrO2 + 4B2O3 → 3ZrB2 + 2ZrO3.
Applications of Zirconium Diboride
Because of its unique properties, zirconium diboride is used in various engineering fields.
Aerospace and aircraft components: The high-temperature capability and light weight of ZrB2 make it suitable for use in aerospace and aircraft components such as turbine blades, vanes, and other high-temperature structural components.
Automotive components: ZrB2 can be used for automotive parts that require high-temperature resistance and lightweight performance, such as exhaust systems and turbochargers.
Electrical and electronic components: The high electrical and thermal conductivity of ZrB2 makes it suitable for use in electrical contacts, electrodes, and heat sinks in electronic components.
Ceramic matrix composites (CMCs): ZrB2 is commonly used as a reinforcing phase in CMCs, which have excellent high-temperature strength and chemical resistance.
Wear-resistant coatings: The high hardness and corrosion resistance of ZrB2 make it an excellent material for wear-resistant coatings in various industrial applications.
Inquiry us